InsightAce Analytic Pvt. Ltd. announces the release of a market assessment report on the “Electronic Investigator Site File (eISF) Market”-, By Deployment Type (Cloud-Based, On-Premise), By End-User (Clinical Research Organizations (CROs), Pharmaceutical and Biotechnology Companies, Academic and Research Institutions, Hospitals and Clinical Sites), By Application (Document Management, Regulatory Compliance, Collaboration Tools, Integration with eTMF/CTMS/EDC), By Component(Software, Services), By Trial Phase (Phase I-IV Clinical Trials, Post-Marketing Surveillance), and Global Forecasts, 2025-2034 And Segment Revenue and Forecast To 2034.”
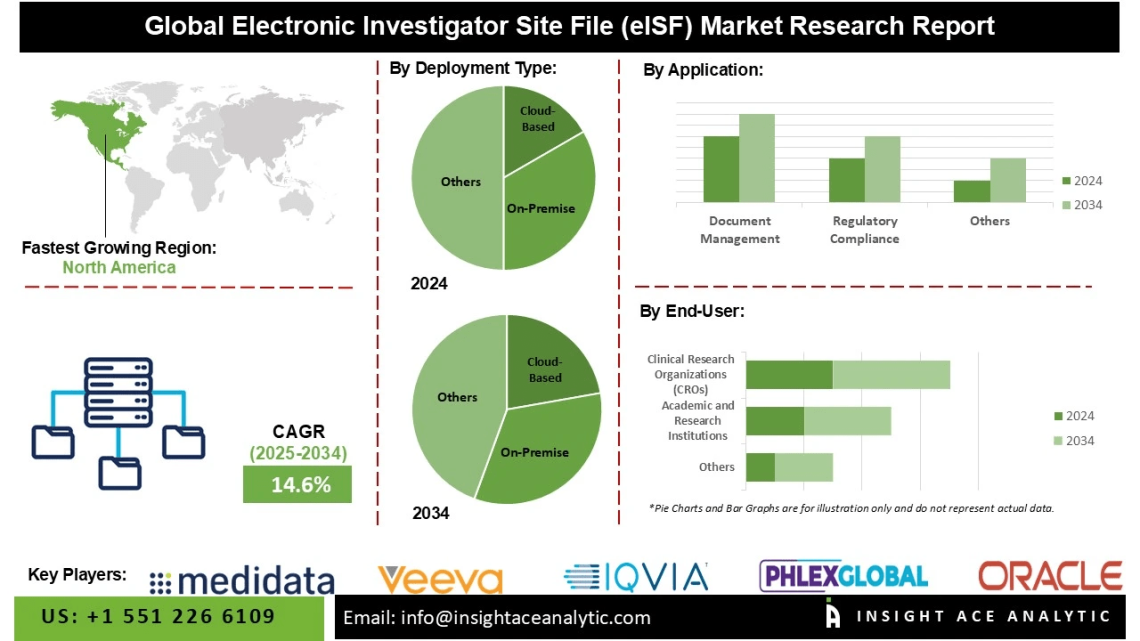
Electronic Investigator Site File (eISF) Market Size is predicted to grow at a 14.6 % CAGR during the forecast period for 2025-2034.
Get Free Access to Demo Report, Excel Pivot and ToC: https://www.insightaceanalytic.com/request-sample/3244
The Electronic Investigator Site File (eISF) functions as the digital equivalent of the conventional Investigator Site File (ISF) in clinical trials, encompassing all essential study-related and regulatory documentation required at trial sites to support monitoring, auditing, inspections, and compliance with Good Clinical Practice (GCP). Key capabilities of eISF systems include electronic signatures, real-time collaboration, version control with comprehensive audit trails, remote access for site personnel, sponsors, and monitors, as well as automated alerts for missing or expiring documents. These features enhance trial preparedness, streamline document management, and reduce administrative overhead.
Market growth for eISF solutions is largely driven by their integration with Electronic Trial Master File (eTMF) platforms, which optimizes the management, compliance, and collaborative handling of clinical trial documentation. Integrated systems provide sponsors, contract research organizations (CROs), and clinical sites with real-time access to critical records, supporting quality assurance, remote monitoring, and expedited decision-making. By minimizing manual document transfers, integration mitigates risks associated with errors, misfiling, and duplication, while automated workflows ensure accurate indexing, synchronization, and audit readiness across eISF and eTMF platforms.
The increasing adoption of standardized, compliant eISF solutions—such as Florence eBinders—has prompted vendors to introduce advanced functionalities, including role-based access controls, dynamic placeholder management, and adherence to international regulatory requirements. Collectively, the integration of eISF and eTMF systems addresses key industry needs, enhancing operational efficiency and reinforcing stakeholder confidence in electronic site file management solutions.
List of Prominent Players in the Electronic Investigator Site File (eISF) Market:
- Veeva Systems
- Medidata Solutions
- Florence Healthcare
- TransPerfect
- Phlexglobal
- ArisGlobal
- Oracle
- IQVIA
- Forte Research
- Ennov
- SureClinical
- MasterControl
- Clinevo Technologies
- RealTime Software Solutions
- Castor EDC
- OpenClinica
- Clinion
- DataTrak
- Medrio
- Cloudbyz
- Fountayn
- Glencoe Software
- StudyTeam
- Agatha Global
Read Overview Report- https://www.insightaceanalytic.com/report/electronic-investigator-site-file-eisf-market/3244
Market Dynamics
Drivers:
The global expansion of clinical trials, fueled by increasing demand for innovative therapies in areas such as oncology, rare diseases, and personalized medicine, has heightened the need for efficient and scalable document management solutions, including electronic Investigator Site Files (eISFs). Digital platforms have become critical for enabling remote monitoring and facilitating seamless collaboration, particularly as decentralized and hybrid trial models gain prominence.
eISFs provide sponsors, contract research organizations (CROs), and clinical sites with real-time, remote access to essential study documents, enhancing transparency, coordination, and the execution of virtual trials. By supporting remote quality control, inspections, and oversight, these systems reduce the reliance on frequent on-site visits. Widespread recognition of these advantages has accelerated adoption among clinical sites, academic institutions, and hospitals, with sponsors further promoting deployment through platform funding, licensing support, and targeted staff training to ensure regulatory compliance and operational efficiency.
Challenges:
Adoption may be constrained by the high costs associated with initial implementation, software licensing, and ongoing system maintenance, particularly for smaller clinical sites or academic institutions with limited budgets and digital infrastructure. Such financial and resource limitations can impede the pace of digital transformation and delay deployment of eISF solutions.
Regional Trends:
North America continues to dominate the market, driven by well-established digital infrastructure and widespread use of clinical trial technologies such as electronic Trial Master Files (eTMFs), Clinical Trial Management Systems (CTMS), and Electronic Data Capture (EDC) platforms. These systems integrate seamlessly with eISF solutions, forming a cohesive digital ecosystem for clinical trial management. The region benefits from a strong regulatory environment, with agencies such as the U.S. Food and Drug Administration (FDA) enforcing compliance with standards including 21 CFR Part 11 for electronic records and signatures. The increasing adoption of decentralized clinical trials (DCTs), coupled with the U.S.’s leading position in the number of clinical trials conducted and its dense network of hospitals, academic medical centers, and private research sites, continues to drive demand for reliable, secure, and remote-access eISF platforms.
Unlock Your GTM Strategy: https://www.insightaceanalytic.com/customization/3244
Recent Developments:
- In July 2024, Clinevo, declared a strategic alliance with Clinion, a top supplier of eClinical solutions. Through this partnership, Clinion’s AI-enabled eClinical platform and Clinevo’s eTMF and Safety Solutions are combined to provide a fully integrated, seamless end-to-end eClinical solution. Clinion now provides Clinevo’s eTMF and Safety Solutions in addition to our current product line as a result of this partnership. Likewise, Clinevo will now be able to provide their clients with Clinion’s full suite of EDC, RTSM, CTMS, ePRO, and eConsent solutions.
- In Sept 2024, ArisGlobal, a cutting-edge life sciences technology business and the developer of LifeSphere®, and Sitero LLC, a supplier of next-generation clinical trial solutions, today announced the extension of their strategic alliance. Through this partnership, ArisGlobal’s LifeSphere R&D technology package incorporates Sitero’s unified eClinical technology platform, which includes Mentor EDC/ePRO, RTSM, eConsent, Payments, and CTMS/eTMF.
Segmentation of Electronic Investigator Site File (eISF) Market.
Global Electronic Investigator Site File (eISF) Market – By Deployment Type
Global Electronic Investigator Site File (eISF) Market – By End-User
- Clinical Research Organizations (CROs)
- Pharmaceutical and Biotechnology Companies
- Academic and Research Institutions
- Hospitals and Clinical Sites
Global Electronic Investigator Site File (eISF) Market – By Application
- Document Management
- Regulatory Compliance
- Collaboration Tools
- Integration with eTMF/CTMS/EDC
Global Electronic Investigator Site File (eISF) Market – By Component
Global Electronic Investigator Site File (eISF) Market – By Trial Phase
- Phase I-IV Clinical Trials
- Post-Marketing Surveillance
Global Electronic Investigator Site File (eISF) Market – By Region
North America-
Europe-
- Germany
- The UK
- France
- Italy
- Spain
- Rest of Europe
Asia-Pacific-
- China
- Japan
- India
- South Korea
- Southeast Asia
- Rest of Asia Pacific
Latin America-
- Brazil
- Argentina
- Mexico
- Rest of Latin America
Middle East & Africa-
- GCC Countries
- South Africa
- Rest of the Middle East and Africa
Why should buy this report:
- To receive a comprehensive analysis of the prospects for the global Electronic Investigator Site File (eISF) Market. To receive an industry overview and future trends of the global Electronic Investigator Site File (eISF) Market
- To analyze the Electronic Investigator Site File (eISF) Market drivers and challenges
- To get information on the Electronic Investigator Site File (eISF) Market. size value (US$ Mn) forecast till 2034
- Major Investments, Mergers & Acquisitions in the global Electronic Investigator Site File (eISF) industry
About Us:
InsightAce Analytic is a market research and consulting firm that enables clients to make strategic decisions. Our qualitative and quantitative market intelligence solutions inform the need for market and competitive intelligence to expand businesses. We help clients gain competitive advantage by identifying untapped markets, exploring new and competing technologies, segmenting potential markets and repositioning products. expertise is in providing syndicated and custom market intelligence reports with an in-depth analysis with key market insights in a timely and cost-effective manner.
Contact us:
InsightAce Analytic Pvt. Ltd.
Visit: https://www.insightaceanalytic.com/
Tel : +1 607 400-7072
Asia: +91 79 72967118
info@insightaceanalytic.com